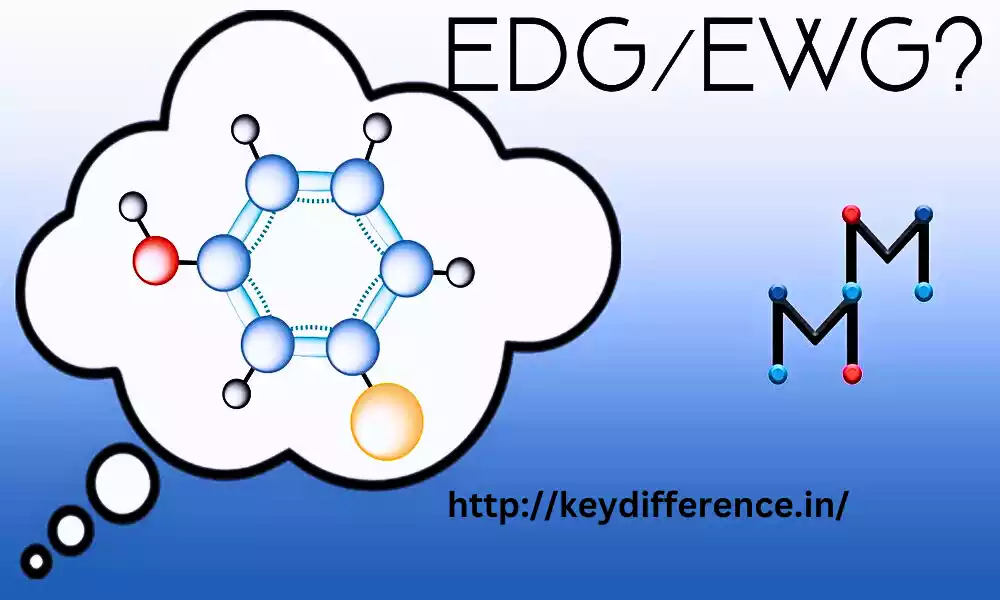
EDG and EWG are two terms that are frequently utilized and traded, driving disarray among numerous people. Both terms hold critical significance in different businesses, and understanding their contrasts is vital for making educated choices. We are going investigate the subtleties between EDG and EWG, shedding light on their applications, benefits, and suggestions in totally different settings. So, let’s jump into the key refinements between EDG and EWG.
Definition of EDG and EWG
EDG (Electron-Donating Group): An Electron-Donating Group (EDG) may be a useful group or molecule that contains a propensity to give or discharge electrons to an atom or a chemical response. An EDG can increment the electron thickness around itself and the neighboring particles, making the atom more nucleophilic or electron-rich. As a result, EDGs generally stabilize positive charges and enhance the reactivity of the molecule towards nucleophilic substitution and addition reactions.
Common causes of EDGs solidify alkyl bunches (e.g., methyl, ethyl), alkoxy bunches (e.g., methoxy, ethoxy), amines (e.g., crucial, collaborator, tertiary amines), and hydroxyl bunches (e.g., -Keen).
EWG (Electron-Withdrawing Group): An Electron-Withdrawing Group (EWG) could be a useful gather or iota that contains a propensity to pull back or draw in electrons from a particle or a chemical response. In natural chemistry, an EWG can diminish the electron thickness around itself and the neighboring iotas, making the atom more electrophilic or electron-deficient. As a result, EWGs generally stabilize negative charges and enhance the reactivity of the molecule towards electrophilic substitution and addition reactions.
Common traces of EWGs solidify carbonyl bunches (e.g., -C=O), nitro bunches (e.g., -NO2), gleaming light (e.g., fluorine, chlorine, bromine, iodine), and cyano bunches (e.g., -CN).
Understanding the electron-donating and electron-withdrawing properties of utilitarian bunches is vital in foreseeing the reactivity and behavior of natural atoms in different chemical responses. These concepts play a noteworthy part in a natural union, sedate plan, and numerous other zones of chemistry.
Importance of understanding EDGs and EWGs in organic chemistry
Understanding Electron-Donating Groups (EDGs) and Electron-Withdrawing Groups (EWGs) is of fundamental significance in natural chemistry for the following reasons:
1. Predicting Reactivity: Information on EDGs and EWGs permits chemists to anticipate the reactivity of natural particles in different chemical responses. EDGs promote nucleophilic attacks and stabilize positive charges, while EWGs promote electrophilic attacks and stabilize negative charges. This predictive ability aids in designing and controlling chemical reactions, leading to the desired products.
2. Reaction Mechanisms: Understanding how EDGs and EWGs influence reaction mechanisms helps in elucidating the pathways and intermediates involved in organic reactions. This knowledge is crucial for mechanistic studies and optimizing reaction conditions.
3. Organic Synthesis: The strategic use of EDGs and EWGs allows chemists to selectively functionalize specific positions on complex molecules. By introducing or removing these groups, chemists can control regioselectivity and stereoselectivity during synthesis.
4. Drug Design: Knowing the effect of EDGs and EWGs on a molecule’s reactivity and properties is vital for drug design. Introducing specific groups can enhance the drug’s bioavailability, target specificity, and metabolic stability.
5. Functional Group Interplay: Many organic molecules contain multiple functional groups, and their interplay can significantly influence the overall reactivity and properties of the compound. Understanding the electronic effects of various functional groups helps predict how they interact in complex systems.
6. Predicting Reactivity of Aromatic Compounds: Aromatic compounds undergo electrophilic aromatic substitution reactions. The presence of EDGs or EWGs on the aromatic ring can determine the regioselectivity of the substitution, guiding the chemist in choosing appropriate reaction conditions.
7. Biological Processes: The understanding of EDGs and EWGs is essential in elucidating enzymatic reactions, molecular interactions, and metabolic pathways.
8. Polymer Chemistry: The reactivity of monomers is influenced by EDGs and EWGs. These impacts play a part in controlling the polymerization handle and deciding the properties of the coming polymer.
9. Material Science: The design and synthesis of new materials often rely on the principles of organic chemistry. Understanding the role of EDGs and EWGs helps in tailoring the electronic and optical properties of materials.
The information on EDGs and EWGs is crucial for understanding the behavior of natural atoms and responses, making it a crucial apparatus for chemists in a wide extend of applications, from medicate improvement to materials science and the past.
Electron-Donating Groups (EDG)
Electron-donating groups (EDGs) are functional groups or atoms in a molecule that tend to donate or release electrons. These groups are electron-rich and increase the electron density around themselves and the neighboring atoms. As a result, EDGs stabilize positive charges and enhance the reactivity of the molecule towards nucleophilic substitution and addition reactions. They can too impact the regioselectivity and stereoselectivity of certain responses. In natural chemistry, the nearness of EDGs can lead to expanded nucleophilicity and the next degree of electron delocalization.
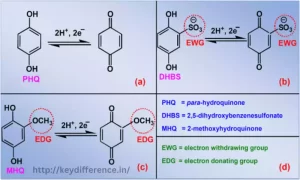
Some common examples of Electron-Donating Groups include:
1. Alkyl groups (e.g., methyl -CH3, ethyl -C2H5)
2. Alkoxy groups (e.g., methoxy -OCH3, ethoxy -OC2H5)
3. Amines (e.g., primary -NH2, secondary -NHR, tertiary -NR2)
4. Hydroxyl groups (e.g., -OH)
EDGs play a significant part in different chemical responses, such as nucleophilic substitution, nucleophilic expansion, and certain radical responses. By increasing electron density, they can stabilize reaction intermediates and transition states, making reactions more favorable and often leading to higher yields.
The nearness of EDGs in natural particles can impact the physical properties of compounds, such as expanding bubbling focuses, softening focuses, and dipole minutes, due to the more grounded intermolecular powers coming about from higher electron thickness.
Understanding the behavior and impacts of Electron-Donating Bunches is fundamental in planning and controlling chemical responses in the natural blend, sedate plan, and other ranges of natural chemistry inquiries. Their key utilization permits chemists to tailor the reactivity and properties of particles, driving the improvement of modern materials and pharmaceuticals with particular wanted characteristics.
Alkyl groups
Alkyl groups are a sort of Electron-Donating Group (EDG) commonly found in natural chemistry. They are useful bunches composed of carbon and hydrogen iotas fortified together in a chain or branched structure. Alkyl groups are characterized by the common equation CnH2n+1, where “n” speaks to the number of carbon molecules within the group.
The only alkyl group is the methyl bunch (-CH3), which comprises a single carbon molecule fortified to three hydrogen iotas. Other diagrams of alkyl bunches connect ethyl (-C2H5), propyl (-C3H7), isopropyl (-C3H7), butyl (-C4H9), and so on.
Key characteristics and properties of alkyl groups include:
1. Electron-Donating Nature: Alkyl groups are considered electron-donating since the carbon-hydrogen bonds in these bunches have a slight positive charge, making the carbon molecule more electrophilic. As a result, alkyl groups can donate electron density to adjacent atoms, making them EDGs.
2. Stability and Inertness: Alkyl groups are generally stable and relatively inert under many reaction conditions, making them versatile building blocks in organic synthesis.
3. Hydrophobicity: Alkyl groups are non-polar and hydrophobic, which influences the dissolvability and behavior of atoms containing these bunches in watery situations.
4. Substitution Reactions: Alkyl bunches take an interest in different substitution responses, such as nucleophilic substitution and radical substitution, due to their capacity to stabilize positive charge through the gift of electron thickness.
5. Isomerism: Alkyl bunches can exist as diverse isomers, such as straight-chain (ordinary) alkyl bunches or branched alkyl bunches, driving basic differences in natural compounds.
6. Alkyl Radicals: Alkyl groups can shape alkyl radicals amid certain chemical responses, which are profoundly receptive species included in radical chain responses.
Alkyl groups are vital components of numerous natural compounds, including alkanes (soaked hydrocarbons), alkyl halides, alcohols, ethers, and more. Their nearness and course of action altogether impact the physical and chemical properties of particles, such as bubbling focuses, softening focuses, and reactivity.
Alkyl groups are widely used in building complex molecules and designing functional materials. Their part in sedate plan and natural frameworks is of awesome significance, as they can impact sedate movement, digestion system, and pharmacokinetic properties.
Electron-withdrawing groups (EWG)
Electron-withdrawing groups (EWGs) are useful bunches or molecules in a particle that incline to pull back or draw in electrons. These groups are electron-deficient and decrease the electron density around themselves and the neighboring atoms. As a result, EWGs stabilize negative charges and enhance the reactivity of the molecule towards electrophilic substitution and addition reactions. They can moreover impact the regioselectivity and stereoselectivity of certain responses. In natural chemistry, the nearness of EWGs can lead to expanded electrophilicity and a better degree of electron localization.

Some common examples of Electron-Withdrawing Groups include:
1. Carbonyl groups (e.g., -C=O)
2. Nitro groups (e.g., -NO2)
3. Halogens (e.g., fluorine -F, chlorine -Cl, bromine -Br, iodine -I)
4. Cyano groups (e.g., -CN)
Each of these groups has an electronegative component (such as oxygen, nitrogen, or incandescent lamp) that draws in electron thickness towards itself, coming about in a mostly positive charge on the carbon or other adjoining iotas.
Key characteristics and properties of Electron-Withdrawing Groups include:
1. Electrophilicity: EWGs make the adjacent carbon or atom more electrophilic, making it susceptible to attack by nucleophiles or electron-rich species.
2. Stabilization of Negative Charges: EWGs stabilize negative charges by delocalizing the excess electron density, making them essential in certain reactions where intermediate anions are involved.
3. Polar Nature: Molecules containing EWGs often exhibit increased polarity due to the unequal sharing of electrons between the atoms involved.
4. Influence on Acidity and Basicity: EWGs stabilize the conjugate base, leading to increased acidity. In bases, they stabilize the conjugate acid, leading to decreased basicity.
5. Reactivity in Electrophilic Aromatic Substitution: EWGs are essential in directing the regioselectivity of electrophilic aromatic substitution reactions in benzene rings.
6. Nucleophilic Addition Reactions: EWGs are included in numerous nucleophilic expansion responses, especially in carbonyl-containing compounds.
7. Bioactivity: The nearness of EWGs in medicate atoms can essentially influence their natural action, digestion system, and poisonous quality.
Understanding the impacts and behavior of Electron-Withdrawing Bunches is significant in anticipating the reactivity of natural atoms in different chemical responses. Their strategic use is instrumental in designing organic synthesis routes, developing pharmaceutical compounds, and tailoring the properties of materials in fields such as polymers and material science.
Carbonyl groups
Carbonyl groups are a particular sort of Electron-Withdrawing Group (EWG) in natural chemistry. They comprise a carbon particle doubly reinforced to an oxygen particle (C=O). The carbon-oxygen twofold bond within the carbonyl group gives critical extremity to the particle, making the carbon particle somewhat positive (electrophilic) and the oxygen particle somewhat negative (nucleophilic).
Carbonyl groups are present in various functional groups, some of which include:
1. Aldehydes: Carbonyl group at the end of a carbon chain (RCHO).
2. Ketones: Carbonyl group in the middle of a carbon chain (RCOR’).
3. Carboxylic acids: Carbonyl group with an attached hydroxyl group (RCOOH).
4. Esters: Carbonyl group with an attached alkoxy group (RCOOR’).
5. Amides: Carbonyl group with an attached amino group (RCO-NR2).
Key characteristics and properties of carbonyl groups incorporate:
1. Electrophilic Nature: The carbon within the carbonyl group is an electrophilic center, making it helpless to be assaulted by nucleophiles.
2. Reactivity in Nucleophilic Addition Reactions: Carbonyl groups promptly experience nucleophilic expansion responses with nucleophiles, driving to the arrangement of alcohols (within the case of aldehydes and ketones), esters, or amides.
3. Influence on Acidity and Basicity: The nearness of a carbonyl bunch in carboxylic acids and amides increases their sharpness compared to other natural compounds. The carbonyl oxygen can stabilize the conjugate base by delocalizing the negative charge.
4. Electrophilic Aromatic Substitution: A carbonyl bunch can act as an EWG and coordinate electrophilic fragrant substitution responses.
5. Hydrogen Bonding: The oxygen within the carbonyl gather can take part in hydrogen holding intuitively, impacting the physical properties and behavior of atoms containing carbonyl bunches.
6. Biological Significance: Carbonyl bunches are predominant in numerous biomolecules, such as sugars, lipids, and amino acids. They play fundamental parts in enzymatic responses and metabolic pathways.
7. Polarity: The nearness of the electronegative oxygen molecule confers critical extremity to the carbonyl group, influencing the dissolvability and intermolecular intuition of atoms.
Carbonyl groups are principal in natural chemistry and have wide applications in different areas, including natural amalgamation, pharmaceuticals, natural chemistry, and materials science. The reactivity and behavior of carbonyl-containing compounds are pivotal to understanding numerous chemical forms and the plan of useful particles and materials.
Difference Between EDG and EWG
The biggest contrast between Electron-Donating Bunches (EDGs) and Electron-Withdrawing Bunches (EWGs) lies in their electron-donating or electron-withdrawing nature and their impacts on electron thickness in natural particles.
Here are the key distinctions between the EDG and EWG:
Electron-Donating Nature (EDG):
• EDGs are functional groups or atoms that tend to donate or release electrons.
• They increase the electron density around themselves and neighboring atoms in a molecule.
• EDGs make the molecule more nucleophilic or electron-rich.
• Common outlines of EDGs consolidate alkyl bunches (e.g., methyl, ethyl), alkoxy bunches (e.g., methoxy, ethoxy), amines, and hydroxyl bunches.
Electron-Withdrawing Nature (EWG):
• EWGs are functional groups or atoms that tend to withdraw or attract electrons.
• They decrease the electron density around themselves and neighboring atoms in a molecule.
• EWGs make the molecule more electrophilic or electron-deficient.
• Common cases of EWGs incorporate carbonyl groups (e.g., -C=O), nitro groups (e.g., -NO2), incandescent lamps (e.g., fluorine, chlorine, bromine, iodine), and cyano groups.
Effect on Reactivity:
• EDGs increase the reactivity of a molecule in nucleophilic substitution and addition reactions. They stabilize positive charges and promote nucleophilic attacks.
• EWGs increase the reactivity of a molecule in electrophilic substitution and addition reactions. They stabilize negative charges and promote electrophilic attacks.
Biological Relevance:
• EDGs are commonly found in biomolecules and can influence enzymatic reactions and metabolic processes.
• EWGs are also present in many drugs and pharmaceutical compounds, where they can affect drug activity, metabolism, and toxicity.
Polar vs. Nonpolar:
• EDGs generally make a molecule more polar due to increased electron density.
• EWGs also increase the polarity of a molecule due to the presence of electronegative elements but to a greater extent.
EDGs give electrons, driving to expanded electron thickness and nucleophilic behavior, whereas EWGs pull back electrons, leading to diminished electron thickness and electrophilic behavior. Understanding the properties and behavior of EDGs and EWGs is significant in foreseeing and controlling the reactivity of natural atoms in different chemical responses and has wide applications in natural unions, medicare plans, and other zones of chemistry.
Electronic Effects on Reaction Mechanisms
Electronic effects play a crucial role in determining reaction mechanisms in organic chemistry. These impacts emerge from the nearness of Electron-Donating Groups (EDGs) and Electron-Withdrawing Groups (EWGs) in atoms. Understanding these impacts is basic for anticipating and clarifying the results of different chemical responses.

Here are some common examples of electronic effects on reaction mechanisms:
1. Nucleophilic Substitution Reactions (SN1 and SN2):
• Where a nucleophile replaces a leaving group, EDGs can stabilize the carbocation intermediate by donating electron density. This leads to increased reaction rates and tends to favor SN1 mechanisms.
• Where a nucleophile attacks a substrate with simultaneous bond breaking, EWGs near the reaction site can destabilize the transition state by withdrawing electron density. This leads to slower reaction rates and tends to favor SN2 mechanisms.
2. Electrophilic Addition Reactions:
• EDGs increase the electron density in the vicinity of the double or triple bond, making the alkene or alkyne more nucleophilic. This enhances the reactivity in electrophilic addition reactions.
• EWGs decrease the electron density in the vicinity of the double or triple bond, making the alkene or alkyne more electrophilic. This also enhances the reactivity in electrophilic addition reactions.
3. Electrophilic Aromatic Substitution (EAS) Reactions:
• EWGs on an aromatic ring increase the electron-withdrawing ability of the ring, making it more susceptible to electrophilic attack. This leads to a higher rate of EAS reactions.
• Conversely, EDGs on the aromatic ring decrease the electron-withdrawing ability, making it less susceptible to electrophilic attack and decreasing the rate of EAS reactions.
4. Benzene Electrophilic Substitution Reactions:
• The electrophile is more attracted to the aromatic ring due to the increased electron deficiency. This enhances the rate of electrophilic substitution.
• Within the nearness of an EDG, the electrophile is less pulled into the fragrant ring, driving a slower rate of electrophilic substitution.
5. Radical Reactions:
• EDGs stabilize radical intermediates by donating electron density, leading to increased rates of radical reactions.
• EWGs can hinder radical reactions by withdrawing electron density, leading to slower rates or inhibition of radical processes.
The electronic impacts on response components are basic in understanding the behavior of particles in chemical responses. By carefully selecting or altering utilitarian bunches in a particle, chemists can control the response results and progress the productivity of engineered forms.
Biological and Pharmaceutical Relevance
The understanding of Electron-Donating Bunches (EDGs) and Electron-Withdrawing Bunches (EWGs) has noteworthy natural and pharmaceutical significance. These electronic impacts play a vital part in the plan, movement, digestion system, and security of drugs and pharmaceutical compounds.
Here’s how EDGs and EWGs are relevant in these fields:
1. Drug Design: EDGs and EWGs are used strategically in drug design to optimize the activity and selectivity of pharmaceutical compounds. By presenting particular utilitarian groups, chemists can fine-tune the electronic properties of a sedate to upgrade its authoritative partiality to the target receptor or protein.
2. Enzymatic Reactions: Enzymes catalyze chemical reactions in living organisms. EDGs and EWGs can impact the reactivity of substrates, influencing the productivity and specificity of enzymatic responses.
3. Pharmacokinetics: The presence of EDGs and EWGs in drug molecules can significantly affect their pharmacokinetic properties, such as absorption, distribution, metabolism, and excretion (ADME). These electronic impacts can affect a drug’s bioavailability and half-life within the body.
4. Toxicity and Side Effects: EDGs and EWGs can influence a drug’s toxicity and potential side effects. Certain utilitarian bunches may be more likely to cause unfavorable responses due to their electronic intuition with natural atoms.
5. Metabolism: EDGs and EWGs in drug molecules can affect their metabolism by interacting with enzymes involved in drug metabolism. This may lead to modified rates of the digestion system and affect a drug’s term of activity.
6. Drug-Drug Interactions: When multiple drugs are administered concurrently, EDGs and EWGs can affect drug-drug interactions. The nearness of one medication may impact the digestion system or adequacy of another sedate within the body.
7. Prodrugs: EDGs and EWGs can be used in prodrug design, where a biologically inactive compound is modified with a functional group to enhance its bioavailability or stability. The functional group is later removed from the body to activate the drug.
8. Target Specificity: EDGs and EWGs can impact a drug’s ability to target specific receptors or enzymes. The nearness of these bunches can progress the selectivity of a sedate for its aiming target while lessening off-target impacts.
9. ADME Optimization: Medicinal chemists use knowledge of EDGs and EWGs to optimize the ADME properties of drug candidates. By controlling the electronic properties, they can make strides in medicate assimilation, dispersion to target tissues, digestion system, and excretion.
Understanding the electronic impacts of utilitarian bunches is basic in medicating disclosure and improvement. It permits pharmaceutical analysts to plan more successful, particular, and secure drugs that can give way better helpful results with decreased side impacts.
Conclusion
Understanding the Difference Between EDG and EWG is vital in comprehending their unique roles in different industries. EDG serves as a portal to outside information, engaging businesses with profitable bits of knowledge for data-driven decision-making. EWG acts as a reliable heat transfer fluid, ensuring optimal performance and protection of machinery in various industrial processes.
By tackling the control of EDG and EWG, businesses can upgrade their operations, accomplish way better proficiency, and make educated choices. So, the other time you come over these acronyms, you’ll have a clear understanding of their particular applications and commitments within the individual spaces.