Introduction of Isoprenoid and Steroid
Organic compounds cover an expansive and varied landscape, including thousands of molecules with diverse structures and functions. Two such classes of molecules that stand out amongst their peers as showing distinct features and having important biological roles include isoprenoids and steroids; understanding their differences and individual roles within biochemistry, pharmacology, and medicine is paramount in understanding their importance for everyday life.
Isoprenoids, also referred to as terpenoids, are organic compounds derived from isoprene with five carbon atoms that serve as essential building blocks in the creation of numerous natural products from pigments and antioxidants to signaling molecules and complex secondary metabolites. Isoprenoids can be found abundantly throughout plants, microorganisms, and animals alike and play essential roles in many vital biological processes.
On the other hand, steroids can be identified by their unique molecular structure known as a steroid nucleus consisting of four fused carbon rings. Steroids serve a number of roles in human, animal, and plant biology such as hormone production or cell membrane components; their most well-known function being hormone regulation in metabolism, growth, and reproduction processes; while cholesterol (a type of steroid) serves a vital function as part of cell membranes in maintaining integrity and fluidity.
Although isoprenoids and steroids exhibit distinct structures and functions, it’s crucial to explore each compound’s distinct features and roles to gain a full appreciation of their significance. This content outline seeks to offer a comprehensive overview of isoprenoids vs steroids differences such as their structures, biosynthesis processes, biosynthesis functions, medical significance as well as potential applications across fields of study. By delving deeper into each class of compounds’ specificities we gain deeper insights into their applications in various domains of science.
Definition of Isoprenoid
Isoprenoids (also referred to as terpenoids) are an organic class of Molecules derived from isoprene (C5H8) Molecules. Isoprene units serve as Building blocks in Producing various isoprenoids found in plants, microorganisms, and animals.
Redox-active compounds play a crucial role in biological systems, serving as pigments, antioxidants, signaling molecules, and precursors for producing complex natural products like essential oils, hormones, and vitamins.
Isoprenoids exhibit a diverse array of chemical structures and biological activities, making them indispensable in fields such as medicine, agriculture, and industry. Examples of isoprenoids include carotenoids, chlorophyll, retinoids rubber as well as essential plant oils.
Definition of Steroid
Steroids are organic compounds distinguished by a specific molecular structure called the “steroid nucleus,” composed of four fused carbon rings arranged as three cyclohexane rings and one cyclopentane ring.
Steroids are widely present in nature and serve various biological purposes; many natural steroids produced in organisms as well as synthetic steroids developed for medical and nonmedical uses have also been synthesized.
Steroids play a critical role in biology as Hormones and cell membrane Components, playing key roles in regulating numerous Physiological processes in humans, Animals, and plants alike. Steroid hormones act as chemical messengers influencing metabolism, growth, reproduction, and immune responses – among them cortisol, estrogen progesterone testosterone aldosterone are examples.
Steroids, in particular Cholesterol, are an integral Component of cell membranes and play an integral role in Maintaining fluidity and Stability, as well as serving as an Intermediary in synthesizing other steroids (hormones).
Steroids play a variety of functions in medicine beyond biological ones, from managing diseases and disorders to treating various conditions. Anti-inflammatory, immunosuppressive, and anti-allergic properties make steroids especially valuable tools in managing diseases and disorders; however, misusing or abusing certain anabolic steroids such as Dbol can have harmful health impacts that should be considered carefully before being misused for medicinal use. Steroids represent a diverse class of compounds with diverse structures and functions that play an integral part in biology, medicine, and physiology.
Comparison Table of Isoprenoid and Steroid
Sure! Here’s a comparison table highlighting the key differences between Isoprenoids and Steroids:
| Aspect | Isoprenoids | Steroids |
|---|---|---|
| Definition | Organic compounds derived from isoprene units | Organic compounds with a steroid nucleus |
| Structure | Varied structures, based on isoprene units | Consists of four fused carbon rings |
| Molecular Formula | Varies depending on the specific isoprenoid | Varies depending on the specific steroid |
| Examples | Carotenoids, chlorophyll, essential oils | Cholesterol, cortisol, estrogen, testosterone, progesterone |
| Biosynthesis | Mevalonate pathway, non-mevalonate pathway | Synthesized from cholesterol or other precursors |
| Biological Functions | Pigments, antioxidants, signaling molecules | Hormones, cell membrane components |
| Occurrence | Abundant in plants, microorganisms, some animals | Present in humans, animals, and plants |
| Medical Significance | Therapeutic potential in herbal medicine | Clinical use in various medical conditions |
| Importance in Industry | Used in the production of natural products | Synthetic steroids developed for various purposes |
| Chemical Properties | Varied chemical properties based on specific isoprenoid | Diverse chemical properties based on specific steroid |
| Side Effects | Minimal side effects observed in natural isoprenoids | Steroid-based medications can have significant side effects |
Please note that this is a simplified comparison table and there may be additional details and nuances beyond what is mentioned here.
Importance of understanding the difference between Isoprenoid and Steroid
Understanding the difference between isoprenoids and steroids is vital, for several reasons:
Isoprenoids and steroids play unique biological roles. Isoprenoids play an integral part in various metabolic processes, acting as pigments, antioxidants, and signaling molecules; contributing to natural products such as essential oils; as well as possessing therapeutic potential in herbal medicine.
Steroids on the other hand play vital roles as hormones and cell membrane components, regulating physiological processes within humans, animals, and plants alike – understanding these special functions helps us gain an insight into their impactful biological impact.
Medical Applications: Isoprenoids and steroids have extensive medical applications. Studies on isoprenoid-based compounds have explored their potential as anti-inflammatory and anticancer agents; natural isoprenoid compounds provide drugs and natural products with therapeutic properties as well as source drugs; synthetic steroids are frequently prescribed to treat inflammation, immune disorders, and hormonal imbalances; knowledge of differences between isoprenoids and steroids can assist with creating targeted therapeutic interventions and understanding potential side effects.
Chemical and Structural Diversity: Isoprenoids and steroids display diverse chemical structures and properties. Isoprenoids cover an expansive spectrum of compounds with various molecular structures, providing rich material for drug development. Steroids boast unique steroid nuclei that influence biological activity. Understanding their structural and chemical differences is vital in studying interactions, synthesis processes, and potential applications across fields as well as interactions and applications in various fields.
Industry and Agriculture: Isoprenoids and steroids play an essential role in several industrial sectors. Their applications range from natural flavorings and fragrances, pharmaceuticals, agricultural applications such as fruit aromatization and flavor enhancement, and plastic production to drug synthesis – isoprenoids are utilized across these fields, thus optimizing their utilization by industry. Steroids (both natural and synthetic) play their part as well. Discerning between isoprenoids and steroids is crucial to ensure optimal utilization across these fields.
Research and Development: Understanding the differences between isoprenoids and steroids provides a basis for further research and development in various disciplines. Exploring their biosynthesis, metabolic pathways, and functions can lead to discoveries of new compounds, therapeutic targets or innovative applications; distinguishing isoprenoids from steroids is crucial in furthering knowledge in biochemistry, pharmacology, plant biology or drug development fields.
Understanding the distinctions between isoprenoids and steroids is vital in understanding their unique properties, functions, and applications in various scientific and industrial domains. Doing so allows researchers, healthcare providers, and industry experts to make more informed decisions, create targeted interventions, and take full advantage of the potential of these compound classes for the improvement of human health, agriculture, and technology.
Isoprene units and their role in isoprenoid formation
Isoprene units play a critical role in the formation and synthesis of isoprenoids. These molecules are formed by joining multiple isoprene units together; most frequently isopentenyl diphosphate (IPP) and dimethylallyl diphosphate (DMAPP), both consisting of five carbon atoms, are used.
Isoprene units serve as building blocks in the creation of isoprenoids. By linking different isoprene units together in different ways, the length and branching patterns of carbon chains within isoprenoids can be adjusted; leading to their wide array of structures found among this class of compounds.
The assembly of isoprenoids occurs through two primary biosynthetic pathways:
Mevalonate Pathway: This pathway serves as the primary method for isoprenoid biosynthesis in animals, fungi, and some bacteria. Isoprene units are produced through the condensation of three molecules of acetyl-CoA into isoprene units to produce malonic acid, which then converts to IPP or DMAPP precursor molecules which then serve as precursors for isoprenoid synthesis.
Non-mevalonate Pathway (Methylerythritol Phosphate Pathway): The non-mevalonate pathway (or Methylerythritol Phosphate Pathway) is another route for isoprenoid biosynthesis found in plants, algae, and some bacteria. Here the precursor molecules glyceraldehyde 3-phosphate and pyruvate undergo several enzymatic reactions to produce IPP and DMAPP respectively.
Once IPP and DMAPP have been synthesized, enzymes can use them to synthesize an array of isoprenoids through condensation reactions, cyclization, modifications, or modification through various methods such as condensation reactions. These processes produce various classes of isoprenoids such as monoterpenes, diterpenes, sesquiterpenes, triterpenes, and polyisoprenes. Isoprene units provide the core building blocks necessary for isoprenoid assembly; their incorporation into larger molecules through enzyme-catalyzed reactions gives this class of compounds its unique structural diversity.
Non-mevalonate pathway (also known as the methylerythritol phosphate pathway)
The non-mevalonate pathway also referred to as the methylerythritol phosphate (MEP) pathway or 2-C-methyl-D-erythritol 4-phosphate (2CmDOP/DOXP) pathway, is an alternative metabolic route for isoprenoid biosynthesis that occurs in various organisms including plants, algae, certain bacteria and apicomplexan parasites. It stands in stark contrast to the mevalonate pathway which serves as the main method for isoprenoid biosynthesis in animals, fungi, and some bacteria.
The non-Mevalonate pathway involves several Enzymatic steps that convert Glyceraldehyde 3-phosphate (GAP) and pyruvate into Isopentenyl diphosphate (IPP) and dimethylallyl Diphosphate (DMAPP), key precursors for isoprenoid Biosynthesis. This pathway operates within plant cells as well as parasitic organisms like Apicomplexan parasites.
Here is a breakdown of the key steps involved in the non-mevalonate pathway:
Step 1: Condensation of GAP and Pyruvate
1. 1-deoxy-D-xylulose 5-phosphate synthase (DXS). The reaction between GAP and pyruvate condenses into 1-deoxy-D-xylulose 5-phosphate (DXP). Step
2. Convert DXP to 2-C-methyl-D-erythritol 4-phosphate (MEP).
Enzyme: DXP Reductase or IspC
Reaction of MEP Kinase (MEPK) with MEP to Form 4-diphosphocytidyl-2-C-methyl-D-erythritol (4DPME).
Step 4 Convert CDPME into 4-diphosphocytidyl-2-C-methyl-D-erythrose (4DP-MEP).
Steps are: 1- CDP-ME 2-phosphatase (CMK or IspF)
Reaction: CDP is dephosphorylated to form CDP-MEP, Step 5- Eliminate MEP from IPP and DMAPP
Enzymes: Multiple enzymes were identified, such as 2-C-methyl-D-erythritol 2,4-cyclodiphosphate synthase (MCT), 4-hydroxy-3-methylbut-2-enyl diphosphate synthase (HDS or IspG), and 4-hydroxy-3-methylbut-2-enyl diphosphate reductase (HDR or IspH).
Reactions: CDP-MEP undergoes various reactions, such as elimination and cyclization, to form IPP and DMAPP from CDP-MEP, which then can be utilized by non-mevalonate pathways for the production of various isoprenoids such as carotenoids, chlorophylls, and essential oils.
The non-mevalonate pathway is an essential route for isoprenoid biosynthesis in many organisms, particularly plants. Understanding its function is crucial to both understanding the biosynthesis of isoprenoids as well as devising strategies to control isoprenoid production in agriculture, medicine, and biotechnology.
Modification of cholesterol to form various steroid hormones
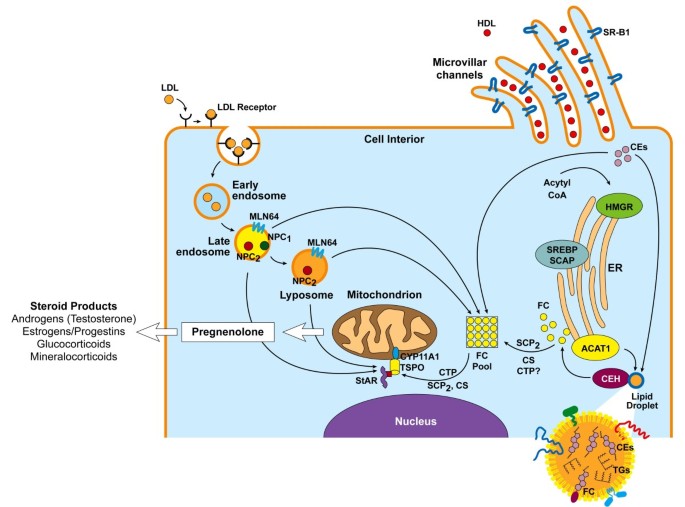
Cholesterol, a form of steroid hormones, serves as the precursor for creating many different kinds of steroid hormones. These hormones are made through various enzymatic processes occurring within specific organs or tissues and derived by cholesterol itself through various modification steps. Here is an outline of this conversion process from cholesterol to various steroid hormones:
Cholesterol as a Base: Cholesterol is an organic molecule synthesized primarily by the liver and also obtained through diet. It serves as an initial substrate for synthesizing steroid hormones.
Converting Cholesterol to Pregnenolone: The first enzymatic step involves the conversion of cholesterol to pregnenolone by mitochondria of steroidogenic cells primarily found in adrenal glands and gonads, using cholesterol side-chain cleavage enzyme (CYP11A1 or P450scc). Pregnenolone serves as a precursor to various other steroid hormones produced during subsequent steps of biosynthesis.
Divergent Paths for Different Steroid Hormones:
Corticosteroids:
Pregnenolone can be converted by adrenal glands into corticosteroids that regulate different physiological processes. Their production involves specific enzymes and steps depending on which corticosteroid type it is produced – for example, cortisol, aldosterone, or corticosterone are just some examples.
Pregnenolone can be converted to androgens like testosterone through a series of enzymatic reactions in the gonads (testes in males and ovaries in females), with 17a-hydroxylase (CYP17A1) and 17b-hydroxysteroid dehydrogenase (HSD17B3) being key enzymes involved in this process.
Pregnenolone can also be converted to estrogens through metabolism in various tissues like the ovaries, placenta, and adipose tissue. Aromatase (CYP19A1) plays an integral part in this process and converts androgens to estrogens for the production of this compound.
Enzymatic modifications and conversions of cholesterol to various steroid hormones occur tissue-specifically and are controlled by various factors, including hormonal signals and feedback mechanisms.
Note that the specific pathways and enzymes involved in steroid hormone synthesis depend on which tissue or organ is being involved, making regulation of steroidogenesis a complex process. In addition, individual steroid hormones may undergo further modification and metabolism in order to exert specific biological effects.
Understanding how cholesterol can be modified into various steroid hormones is fundamental for understanding endocrine pathways, reproductive processes and the various functions played by steroid hormones in physiological functions.
Functions as pigments, antioxidants, and signaling molecules
Isoprenoids serve multiple functions, including pigments, antioxidants, and signaling molecules.
Below is more information regarding each application:
Pigments: Many isoprenoids act as pigments to add hues found in nature, providing bright hues such as those seen on leaves. Examples include:
Carotenoids: Carotenoids are a class of Isoprenoids Responsible for the vibrant red, orange, and Yellow hues found in fruits, Vegetables, and flowers. Carotenoids play an integral part in Photosynthesis by acting as light-absorbing Pigments while simultaneously serving as Antioxidants to protect plants against damage caused by excessive light exposure or reactive oxygen species.
Chlorophylls: Chlorophylls are Isoprenoid pigments found in plants and algae that play an Essential role in Photosynthesis by capturing light energy and turning it into Chemical energy for use by living Things. Chlorophylls give leaves their distinctive green hue while providing essential amino acid chains necessary for synthesizing carbohydrates within plants.
Phycobilins: These isoprenoid-derived pigments can be found in certain algae and cyanobacteria, contributing to their unique red, blue, or purple colors as well as light harvesting during photosynthesis.
Antioxidants: Isoprenoids also have Antioxidant properties, helping protect cells Against damage caused by free radicals and reactive oxygen species. Some of the more notable isoprenoids with this property include:
Vitamin E (Tocopherols & Tocotrienols): Isoprenoid-derived forms of Vitamin E such as Tocopherols & Tocotrienols act as powerful antioxidants that neutralize free radicals while protecting cells against oxidative stress.
Coenzyme (Ubiquinone): Coenzyme is a molecule that occurs in all cells. It plays a vital role in the production of energy and also acts as an antioxidant to protect DNA and cell membranes from damage caused by free radicals.
Signaling Molecules: Isoprenoids have long been recognized as signaling molecules that Contribute to numerous Physiological Processes. For instance, they participate in intercellular communication, regulate gene expression levels, and modulate cell signaling pathways – examples being:
Prenylated Proteins: Isoprenoids such as farnesyl pyrophosphate (FPP) and geranylgeranyl pyrophosphate (GGPP), essential in post-translational modification of proteins, are instrumental for targeting and anchoring proteins to cell membranes; their prenylation helps alter both localization and function of these proteins.
Steroid Hormones: Derived from cholesterol, steroid hormones are isoprenoid-based signaling molecules that modulate various physiological processes. For instance, cortisol, aldosterone, testosterone, and estradiol all serve as signaling molecules regulating metabolism, immune responses reproductive functions as well as other biological processes.
In essence, isoprenoids serve as pigments, antioxidants, and signaling molecules for living organisms. Their vibrant hues add vibrancy to plants while their antioxidative capabilities help shield cells against damage caused by free radicals and their signaling functions play key roles in intercellular communication and physiological process regulation – making isoprenoids integral components of biological systems.
Contribution to the production of natural products such as terpenes and essential oils
Isoprenoids play an Essential role in the production of Natural products, including Terpenes and Essential oils. These Compounds are found Throughout nature’s plant kingdom and give various herbs, spices, fruits, and flowers their characteristic aromas and flavors.
Here is how isoprenoids contribute to these natural products:
Terpenes:
Formation of Basic Building Blocks: Isoprenoids such as isopentenyl diphosphate (IPP) and dimethylallyl diphosphate (DMAPP) serve as essential building blocks in terpene production; these compounds are produced via the mevalonate pathway in plants and certain microorganisms.
Terpene Synthases: Terpene synthases are enzymes that catalyze the assembly of IPP and DMAPP into various terpenes, while simultaneously controlling their specific structure and stereochemistry. Different terpene synthases produce various types of terpenes including monoterpenes, sesquiterpenes, diterpenes, and triterpenes.
Terpenes Perform Multiple Biological Tasks in Plants: Terpenes serve a wide range of biological purposes in plants. They act as attractants for pollinators or seed dispersers, provide protection from herbivores and pathogens, regulate growth and development and even help with regulation. Some well-known terpenes include limonene, pinene, menthol, and geraniol which serve a number of these purposes.
Essential Oils:
a. Terpene Dominance: Essential oils are volatile mixtures of compounds, with terpenes often being the most abundant component; they produce distinctive scents and flavors associated with different plants, contributing aromatic and therapeutic qualities as well as aroma.
b. Extraction and Utilization: Essential oils are extracted from plant materials using various extraction techniques such as steam distillation, cold pressing, or solvent extraction. Their widespread uses span perfumery, aromatherapy, traditional medicine, cosmetics, and food and beverage industries; each essential oil boasts its own specific mix of terpenes and volatile compounds which give rise to its unique aroma as well as potential health benefits.
c. Therapeutic Potential: Essential oils contain many terpenes with therapeutic potential. Some have antibacterial, antifungal, or antiviral activities while others possess analgesic or soothing properties, making these oils especially suitable for complementary and alternative medicine treatments. These properties make essential oils indispensable in healthcare settings that employ alternative and complementary approaches.
Isoprenoids play an integral part in terpene biosynthesis, leading to natural products like terpenes and essential oils. These compounds offer an exquisite variety of aromas, flavors and potential health benefits, enriching our sensory experiences while serving as valuable resources in various industries.
Functions as hormones and cell membrane components
Isoprenoids play an essential role as hormones and cell membrane components, providing support to various biological processes. Here’s a closer look at their Functions:
Hormones:
Steroid Hormones:
Steroid hormones, Composed of Isoprenoid-based compounds derived from Cholesterol, act as signaling molecules within the body. Common examples of such steroid hormones include cortisol, aldosterone, testosterone, and estradiol which all serve to modulate various physiological processes including metabolism, immune responses reproductive functions stress responses by binding to specific receptors on target cells thereby altering gene expression or signaling pathways.
Isoprenoids also serve as plant hormones. Gibberellins and abscisic acid (ABA), both produced from isoprenoids, act as isoprenoid-derived plant hormones to control plant growth and development, such as stem elongation, seed germination, and flowering; while ABA plays an integral part in stress responses such as drought tolerance and dormancy tolerance.
Cell Membrane Components: Cholesterol: Cholesterol is an essential lipid-based component of animal cell membranes that helps maintain fluidity, stability, and permeability while simultaneously shaping their organization and function. Cholesterol plays an especially significant role in plasma membranes where it facilitates signal transduction and intercellular communication processes.
Prenylated Proteins: Isoprenoids such as farnesyl and geranylgeranyl groups can be used to modify proteins through a process known as prenylation. Prenylation involves attaching specific lipid moieties to specific proteins in order to anchor them onto cell membranes and has numerous functions, including signal transduction, protein targeting, and membrane localization.
Coenzyme Q10 (CoQ10), an isoprenoid molecule, is an essential part of mitochondria’s electron transport chain. As such, it plays an essential role in respiration and energy production within cells, as well as participating in ATP synthesis via electron shuttle. Furthermore, CoQ10 serves as an antioxidant by protecting membranes and mitochondria from damage due to oxidation.
Isoprenoids play an indispensable role in maintaining optimal cell functioning, signaling, and organismal homeostasis. Their roles as hormones and cell membrane components play a significant role in maintaining proper cellular functionality and homeostasis while contributing to physiological processes and supporting cell membrane integrity and function.
Therapeutic potential of isoprenoids in herbal medicine
Isoprenoids found abundantly in plants, offer great therapeutic potential in herbal medicine. These natural Compounds offer various health benefits that have been utilized by Traditional healers for centuries – here are a few noteworthy therapeutic uses of Isoprenoids in herbal medicine:
Anti-Inflammatory Effects: Numerous isoprenoids exhibit anti-inflammatory effects, allowing them to help alleviate inflammation-related conditions. Curcumin, an anti-Inflammatory Terpenoid found in Turmeric, has Powerful anti-Inflammatory activity and can relieve Symptoms Associated with arthritis, Inflammatory bowel disease, and other Inflammatory Conditions.
Antioxidant Activity: Isoprenoids typically exhibit antioxidant activity, helping combat oxidative stress and protect cells from damage caused by free radicals. Resveratrol, a Polyphenolic compound derived from plants such as Grapes and berries, exhibits Antioxidant effects that may provide Cardioprotective and anti-aging benefits.
Antimicrobial Action: Certain isoprenoids exhibit antimicrobial action against bacteria, fungi, and parasites. Thymol and carvacrol derived from essential oils of thyme and oregano are two isoprenoids known to show this property and serve as natural alternatives to conventional antimicrobial agents.
Anti-Cancer Activity: Certain isoprenoids have shown promising anti-cancer activity by suppressing tumor growth, inducing apoptosis (programmed cell death), and blocking blood vessel formation that supplies tumors – this includes Paclitaxel from Pacific Yew trees as well as Artemisinin from Artemisia annua (sweet wormwood) used for malaria treatments.
Neuroprotective Properties: Isoprenoids such as terpenes and flavonoids have demonstrated neuroprotective properties, benefiting brain health. Ginkgo biloba extract Containing terpenoids such as Ginkgolides and Bilobalides has long been used to boost cognitive Performance while alleviating Symptoms associated with Neurodegenerative disorders.
Cardiovascular Health: Numerous isoprenoids have been found to contribute to cardiovascular wellness by lowering cholesterol levels, improving blood circulation, and strengthening heart function. Garlic contains sulfur-containing compounds called allicin that have cardioprotective effects by lowering both blood pressure and cholesterol levels.
Digestive Support: Isoprenoids found in herbs such as peppermint, such as menthol, and menthone, may provide digestive relief by soothing digestive discomfort, alleviating symptoms associated with Irritable Bowel Syndrome (IBS), and supporting healthy digestion.
Anti-Anxiety and Mood-Enhancing Effects: Certain Isoprenoids found in lavender and citrus fruit such as Linalool have anti-anxiety and mood-enhancer Properties that make Aromatherapy effective at relieving anxiety and stress Reduction. Aromatherapy utilizes these compounds as therapeutic tools for relaxation and stress reduction.
Note that isoprenoids’ therapeutic potential depends heavily on factors like dosage, formulation, and individual variations; therefore herbal medicine practitioners and healthcare professionals should always be consulted prior to using products with isoprenoid-rich herbal ingredients.
Conclusion
Understanding the distinctions between isoprenoids and steroids is critical to understanding their distinct properties and biological roles. Isoprenoids, derived from isoprene units, cover a broad spectrum of compounds serving as pigments, antioxidants, signaling molecules, and building blocks for natural products like terpenes and essential oils. On the other hand, steroids comprise a specific class of four-ring compounds used as hormones or components of cell membranes – as such their differences must be clearly understood for proper understanding and evaluation.