Introduction
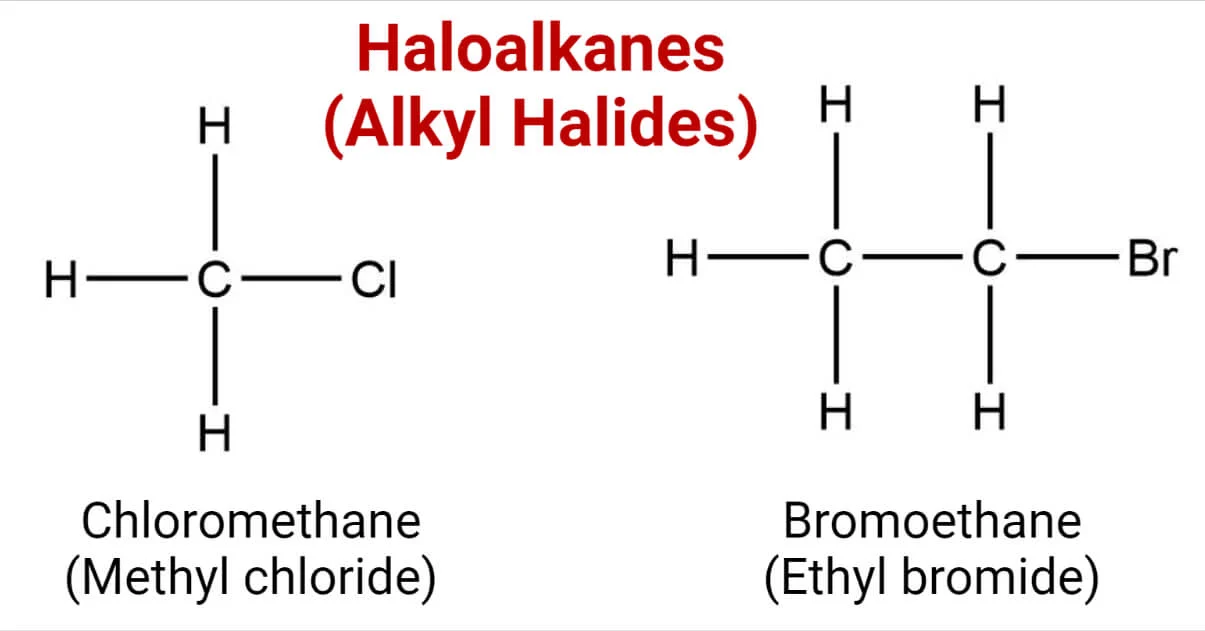
Halogenoalkanes, Commonly referred to as alkyl halides or Haloalkanes, are organic Compounds containing one or more Halogen atoms bonded to carbon atoms, such as fluorine, chlorine, Bromine or iodine. They play a significant role in organic chemistry due to their varied chemical reactivity and wide array of applications across industries.
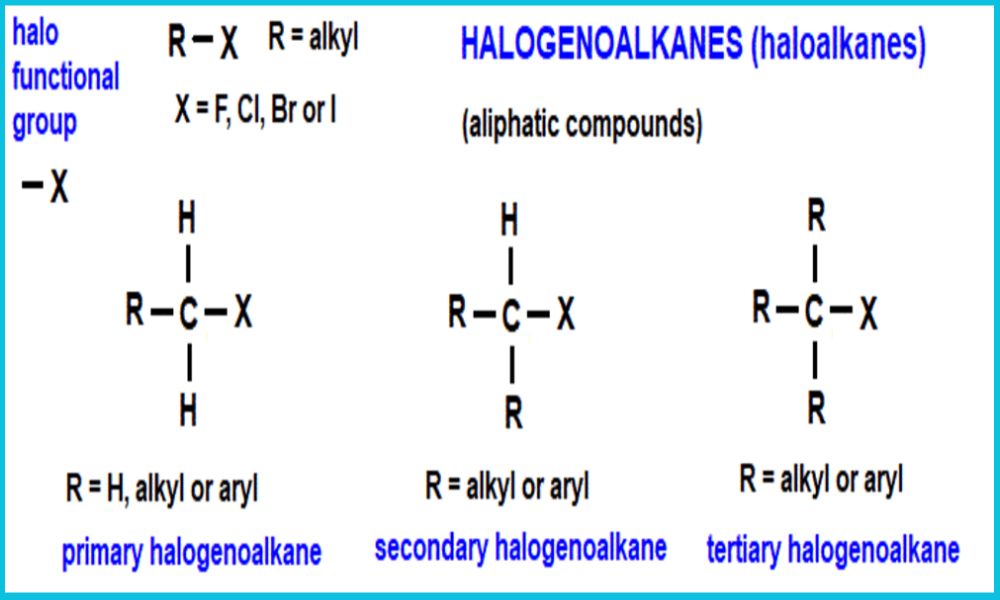
Understanding halogenoalkanes requires understanding their structure. In particular, distinguishing among primary, secondary, and tertiary halogenoalkanes based on how many alkyl groups attach themselves to carbon atoms carrying the halogen atom is of great significance in understanding this field of research.
Primary Halogenoalkanes contain one alkyl group attached to their carbon atom Containing the halogen; secondary and tertiary Halogenoalkanes contain two such bonds; finally tertiary Halogenoalkanes boast three alkyl groups Bonded directly onto this Carbon.
Understanding these three types of halogenoalkanes has an enormous effect on their chemical properties, reactions and applications. Being aware of their differences allows one to more accurately predict and manipulate how these materials behave in various reactions such as nucleophilic substitutions, elimination reactions or organic transformations.
This content outline will examine the definition, structure, preparation methods, chemical properties and applications of primary, secondary, and tertiary halogenoalkanes.
By understanding their unique properties we can gain an understanding of their reactivity patterns as well as understand their practical applications in diverse fields such as pharmaceuticals, agrochemicals, materials science and environmental chemistry.
Definition of Primary Halogenoalkanes
Primary halogenoalkanes, also referred to as primary alkyl halides, are organic compounds composed of two carbon atoms linked by one single carbon bond directly connected with one or more halogen atoms bonded directly together; this bonding between carbons is known as primary carbon bonding and its primary carbon atom is known as primary carbon atom.
Their general formula for primary halogenoalkanes includes RCH2X where R represents alkyl or aryl groups and X represents their respective halogen atom (either fluorine chlorine bromine or iodine).
Primary halogenoalkanes can be identified by their presence of one alkyl group attached to the carbon atom bearing the halogen, which affects their reactivity and chemical properties in comparison to secondary and tertiary halogenoalkanes. Furthermore, primary carbons provide less steric hindrance during reactions allowing nucleophiles easier access to them for chemical reactions.
Primary halogenoalkanes can be synthesized using various methods, including substitution reactions in which a halogen atom replaces a functional group attached to carbon atoms. These compounds find use in organic synthesis, pharmaceutical production and other chemical processes requiring the introduction of specific functional groups.
Note that primary halogenoalkanes tend to exhibit higher reactivity and undergo nucleophilic substitution reactions more readily compared to secondary and tertiary halogenoalkanes, necessitating more rigorous handling and safety precautions when working with these substances, as they can be toxic and have environmental ramifications if mishandled.
Definition of Secondary Halogenoalkanes
Secondary halogenoalkanes, also referred to as secondary alkyl halides, are organic compounds composed of two carbon atoms bound together via covalent bonds with one or more halogen atoms bonded directly. The carbon directly connected to the halogen atom is known as its secondary carbon atom; their general formula for secondary halogenoalkanes is R2CX where R represents alkyl or aryl groups while X refers to fluorine chlorine bromine or iodine).
Secondary halogenoalkanes can be identified by two alkyl groups attached to a carbon atom bearing halogens, creating different chemical properties compared to primary and tertiary halogenoalkanes. Furthermore, this structural arrangement results in different reactivity and chemical properties than their primary/tertiary counterparts due to potential steric hindrance effects that alter reaction rates and selectivities of different chemical reactions.
Preparing secondary halogenoalkanes typically requires substitution reactions in which a halogen atom replaces one of the functional groups attached to secondary carbon. These compounds find use across many fields such as organic synthesis, pharmaceuticals and solvents due to their reactive nature and ability to introduce specific functional groups into molecules. It is important that secondary halogenoalkanes be handled carefully as their formation can have toxic and environmental consequences if mismanaged.
Definition of Tertiary Halogenoalkanes
Tertiary halogenoalkanes, also referred to as tertiary alkyl halides, are a form of halogenoalkanes in which the carbon atom bonded to an halogen (e.g. fluorine, chlorine, bromine or iodine) is directly attached to three alkyl groups – making the carbon carrying this element “tert”-in nature.
Structure-wise, tertiary halogenoalkanes can be represented as R3CX in their structural formula; R denotes an alkyl group such as methyl, ethyl or propyl and X represents the halogen atom. Any number of alkyl groups bonded to carbon carrying this halogen could exist within its structures; all may share it.
Tertiary halogenoalkanes contain three alkyl groups bound to their carbon atom, making them highly branched and sterically hindered, which impacts their reactivity and stability compared to primary and secondary halogenoalkanes.
Due to their bulky alkyl groups, tertiary halogenoalkanes tend to exhibit different chemical properties and reactivity patterns than primary and secondary halogenoalkanes. They typically exhibit less nucleophilic substitution reactions and more frequently undergo elimination reactions such as E1 or E2 reactions in which one or more halides ions are eliminated simultaneously with the formation of double bonds or cyclic compounds.
Tertiary halogenoalkanes find applications across several fields, such as organic synthesis, pharmaceuticals and chemical industry. Their unique combination of reactiveness and stability makes them useful starting materials for creating complex organic molecules.
Comparison Table of Primary Secondary and Tertiary Halogenoalkanes
Sure! Here’s a comparison table outlining the key differences between primary, secondary, and tertiary halogenoalkanes:
| Characteristics | Primary Halogenoalkanes | Secondary Halogenoalkanes | Tertiary Halogenoalkanes |
|---|---|---|---|
| Structure | Halogen attached to a carbon bonded to one alkyl group | Halogen attached to a carbon bonded to two alkyl groups | Halogen attached to a carbon bonded to three alkyl groups |
| Reactivity with nucleophiles | More reactive towards nucleophilic substitution reactions | Moderately reactive towards nucleophilic substitution reactions | Less reactive towards nucleophilic substitution reactions |
| Substitution reactions | Tends to undergo substitution reactions more readily | Undergoes substitution reactions at a moderate rate | Undergoes substitution reactions at a slower rate |
| Elimination reactions | Less prone to elimination reactions | Can undergo elimination reactions in addition to substitution reactions | More prone to elimination reactions |
| Steric hindrance | Least steric hindrance due to fewer alkyl groups | Moderate steric hindrance due to two alkyl groups | Significant steric hindrance due to three alkyl groups |
| Stability | Relatively stable | Moderately stable | Less stable due to increased steric hindrance |
| Examples | Methyl bromide (CH3Br), Ethyl chloride (CH3CH2Cl) | Isopropyl bromide ((CH3)2CHBr), Isobutyl chloride ((CH3)2CHCH2Cl) | Tert-butyl bromide ((CH3)3CBr), Tert-amyl chloride ((CH3)3CCl) |
| Applications | Used as intermediates in organic synthesis, solvents, and in pharmaceutical and agrochemical industries | Employed in synthesis of complex organic compounds, pharmaceuticals, and as reagents in various reactions | Used as starting materials in organic synthesis, catalysts, and in the production of polymers |
Please note that this table provides a general comparison and the characteristics mentioned can vary depending on specific compounds within each category of halogenoalkanes.
Importance of understanding different types of halogenoalkanes

Understanding the various types of halogenoalkanes – primary, secondary, and tertiary halogenoalkanes – is of crucial importance for various reasons:
Reactivity and Chemical Properties: The type of halogenoalkane greatly determines its reactivity and chemical properties. Primary, secondary, and tertiary halogenoalkanes each display distinct patterns due to the number of alkyl groups attached to a carbon bearing a halogen atom; understanding these variations allows you to accurately predict their behavior in various reactions such as nucleophilic substitutions and elimination reactions.
Reaction Mechanisms: Halogenoalkane reactivity is determined by their unique reaction mechanisms. Primary, secondary, and tertiary halogenoalkanes use different nucleophilic substitution and elimination processes that influence reaction outcomes. Understanding these reactions is integral for designing synthetic routes as well as understanding factors affecting reactions outcomes.
Synthetic Applications: Halogenoalkanes serve as versatile starting materials in organic synthesis. Different types of halogenoalkanes may be better suited for certain transformations due to their different degrees of reactivity or ease of functional group manipulation. Being familiar with primary, secondary, and tertiary halogenoalkanes enables chemists to select suitable reactants and develop efficient synthetic pathways.
Pharmaceutical and Agrochemical Industries: Halogenoalkanes play an integral part in the creation of pharmaceuticals and agrochemicals, with understanding their reactivity and properties being essential to designing drug molecules with optimal bioavailability, control over bioavailability and optimizing pharmacological activity. Furthermore, halogenoalkanes find use as building blocks in their synthesization into agrochemicals which play key roles in crop protection as well as increasing agricultural productivity.
Environment Impact: Halogenoalkanes have potential environmental ramifications due to their potential toxicity and persistence in the environment, so understanding their various forms is critical to understanding their fate, degradation pathways and ecological effects – information necessary for creating effective remediation or pollution control strategies.
Safety Considerations: Obtaining an understanding of the properties and reactions associated with different halogenoalkanes is vital to safely handling them in lab and industrial settings. Their potential hazards, including flammability, toxicity, and reactivity vary based on their type. Therefore, appropriate knowledge and precautions must be taken for safe handling, storage and disposal of halogenoalkanes.
Understanding halogenoalkanes is vital to successfully and responsibly using them in scientific and industrial applications, from predicting their reactivity, designing synthetic routes, developing pharmaceuticals and agrochemicals, assessing environmental impacts and assuring safety during handling processes. Understanding their different forms helps provide the basis for efficient utilization.
Reactivity towards nucleophilic substitution reactions
Nucleophilic substitution reactions involving halogenoalkanes are key reactions, where a nucleophile replaces the halogen atom with another nucleophile – this reaction typically distinguishes primary, secondary and tertiary halogenoalkanes in terms of their relative reactivity in these reactions.
A. Halogenoalkanes of Primary Interest:
Reactivity: Halogenoalkanes are highly reactive towards nucleophilic substitution reactions. Within these reactions lies an SN2 mechanism (substitution nucleophilic bimolecular).
Transition State: When in transition state, nucleophile attacks the carbon atom directly opposite from where the leaving halogen atom has left, leading to simultaneous bond formation and bond-breaking processes.
Steric Hindrance: Due to their absence of alkyl groups, primary halogenoalkanes provide less steric hindrance for nucleophilic approaches than secondary and tertiary types of halogenoalkanes.
Rate of Reaction: Primary Halogenoalkanes have the fastest reaction rates among these three categories of compounds.
B. Secondary Halogenoalkanes
Reactivity: Secondary halogenoalkanes have moderate nucleophilic substitution reactivity.
Mechanism: This reaction can proceed via both SN2 (substitution nucleophilic unimolecular) and SN1 (substitution nucleophilic unimolecular) mechanisms depending on reaction conditions.
Transition State: For transition states in which nucleophiles attack opposite of leaving groups (like primary halogenoalkanes), two pathways exist. For the SN2 path, nucleophiles attack directly the carbon atom opposite leaving groups (similar to primary halogenoalkanes).
While for the SN1 pathway dissociation first takes place prior to attack from nucleophiles; when followed by dissociation by leaving groups followed by carbocation intermediate which then attacks nucleophiles ( like primary halogenoalkanes).
Steric hindrance: Secondary halogenoalkanes exhibit moderate steric hindrance due to the presence of one alkyl group, limiting access to their nucleophile. Rate of Reaction: Secondary halogenoalkanes typically exhibit slower reaction rates compared to primary ones.
C. Tertiary Halogenoalkanes:
Reactivity: Tertiary halogenoalkanes exhibit low nucleophilic substitution reactions.
Mechanism: This reaction predominantly proceeds via the SN1 mechanism.
Transition State: Tertiary halogenoalkanes form stable carbocation intermediates through dissociation of their leaving groups, before nucleophiles attack this carbocation intermediate and form product.
Steric Hindrance: Tertiary Halogenoalkanes experience significant steric hindrance due to their three alkyl groups, making it challenging for nucleophiles to approach the reaction site.
Rate of Reaction: Tertiary Halogenoalkanes have one of the slowest reactions rates among the three types.
Understanding nucleophilic substitution reactions is crucial in organic synthesis and for selecting reaction conditions to produce desired products.
Stability and ease of formation of carbocation intermediates
Carbocation intermediates play an essential part in certain reactions involving halogenoalkanes. Their stability and ease of formation depend on whether their source halogenoalkane is primary, secondary, or tertiary in its nature.
A. Halogenoalkanes of Primordial Origin:
Stability of Carbocation: Primary carbocations structures are highly unstable due to a lack of alkyl groups that can donate electron density and stabilize positive charges.
Ease of Formation: Primary carbocations formation can be challenging due to insufficient electron-donating groups on carbon that bear positive charges, making synthesis challenging.
B. Secondary Halogenoalkanes:
Stability of Carbocation: Secondary carbocations is more stable than its primary counterpart due to the presence of one alkyl group which can contribute electron density through hyperconjugation and support positive charge stability.
Ease of Formation: Secondary carbocationss can be formed more readily compared to primary ones, yet still remain less stable than their tertiary counterparts. C. Tertiary Halogenoalkanes:
Stability of Carbocation: Tertiary carbocations structures are among the most stable of their three forms due to three alkyl groups that help increase positive charge stabilization via hyperconjugation.
Formation Ease: Tertiary carbocations formation is among the easiest of any of the primary, secondary and tertiary halogenoalkanes due to the greater stability provided by alkyl groups.
Stability and ease of formation of carbocation intermediates have an enormous effect on overall reaction mechanisms and product distribution. Reactions that use carbocations intermediates such as rearrangements and certain substitution reactions benefit greatly when stable carbocation intermediates exist; it is therefore imperative that when predicting outcomes involving halogenoalkanes.
Use of halogenoalkanes in organic synthesis
Halogenoalkanes, more commonly referred to as alkyl halides, are highly versatile compounds used extensively in organic synthesis for various transformations and reactions. With their distinct reactivity and functional groups providing valuable building blocks for producing a wide array of organic compounds. Here are a few applications and uses of halogenoalkanes in organic synthesis:
A. Nucleophilic Substitution Reactions:
SN2 Reactions: Halogenoalkanes can serve as substrates for nucleophilic substitution reactions in which their halogen atom is replaced by a nucleophile, creating new functional groups in an alkyl chain and thus expanding chemical diversity of synthesized compounds.
SN1 Reactions: Tertiary halogenoalkanes undergo nucleophilic substitution reactions using an SN1 mechanism, producing carbocation intermediates. These reactions can be used in the production of complex organic molecules like natural products and pharmaceuticals.
B. Elimination Reactions (E2):
Halogenoalkanes may undergo elimination reactions known as E2 reactions, in which both halogen atoms and proton can be eliminated to create double bonds – these reactions are very useful in synthesizing alkenes as well as producing unsaturated bonds to construct cyclic compounds or introduce unsaturated bonds into compounds.
Cross-Coupling Reactions: Halogenoalkanes such as alkyl bromides or iodides can be used as coupling partners in cross-coupling reactions such as Suzuki, Stille, and Negishi reactions in order to create new carbon-carbon bonds, leading to the synthesis of complex organic molecules as well as functional materials.
Grignard Reactions: Halogenoalkanes can be used as starting materials in Grignard reactions, where metal (e.g. magnesium) reacts with the halogenoalkane to form organomagnesium compounds that serve as versatile precursors for alcohols, ketones, carboxylic acids and other functional groups synthesis.
D. Synthetic Intermediates:
Protective Groups: Halogenoalkanes can serve as protecting groups for specific functional groups in multi-step synthesis processes. By temporarily converting it into a less reactive halogenoalkane form, unwanted reactions can be avoided and selective modifications made at later stages.
Substituents and Functionalization: Halogenoalkanes serve as effective starting materials to introduce specific substituents or functional groups into organic molecules, with subsequent reactions modifying or replacing individual halogen atoms with others that better suit subsequent reactions.
E. Industrial Applications:
Solvents and Extractants: Halogenoalkanes such as chloroform and carbon tetrachloride have many applications as solvents and extractants in various industrial processes.
Refrigeration and Propellants: Chlorofluorocarbons (CFCs) and hydrochlorofluorocarbons (HCFCs) were once widely used as refrigerants and propellants; however, their usage has since been curtailed due to environmental considerations.
Halogenoalkanes provide organic chemists with access to a vast array of compounds with diverse structures and functionalities. Their versatility and reactivity make them indispensable tools in building complex organic molecules as well as developing new materials and pharmaceuticals.
Role of primary, secondary, and tertiary halogenoalkanes in specific reactions
Halogenoalkanes exhibit different chemical reactivity depending on their classification as primary, secondary or tertiary; each type plays its own specific role in various reactions. Understanding these roles is essential in organic synthesis.
Here are examples of primary secondary and tertiary halogenoalkanes being utilized during specific chemical processes:
A. Nucleophilic Substitution Reactions:
SN2 Reactions:
mes mes Its mes Primary halogenoalkanes make them suitable substrates for SN2 reactions, as their low steric hindrance facilitate backside attacks by nucleophiles, resulting in effective substitution reactions.
Secondary Halogenoalkanes: While secondary halogenoalkanes can undergo SN2 reactions, they tend to react more slowly due to increased steric hindrance from alkyl groups in their structure.
Tertiary Halogenoalkanes: Tertiary halogenoalkanes tend to be poor substrates for SN2 reactions due to significant steric hindrance caused by multiple alkyl groups near the reaction center.
SN1 Reactions:
a. Primary Halogenoalkanes:
These types of molecules do not undergo SN1 reactions as readily due to less favorable carbocation intermediate formation conditions.
Secondary Halogenoalkanes: Secondary halogenoalkanes are widely employed in SN1 reactions due to the more stable carbocation intermediate created due to the presence of alkyl groups allowing subsequent nucleophilic attack.
Tertiary Halogenoalkanes: Tertiary halogenoalkanes can easily undergo SN1 reactions as the highly stable tertiary carbocation intermediate can form quickly, leading to successful nucleophilic substitution reactions.
B. Elimination Reactions (E2):
a. Primary Halogenoalkanes: mes Itsalitat facilitate E2 reactions quite efficiently due to little or no steric hindrance. The presence of significant steric hindrance allows proton removal while leaving one halogen atom behind in order to form double bonds in multiples of three elements in total.
b. Secondary Halogenoalkanes: Secondary Halogenoalkanes are particularly reactive in E2 reactions. Their alkyl groups stabilize the transition state for efficient elimination.
c. Tertiary Halogenoalkanes: Tertiary halogenoalkanes can easily undergo E2 reactions due to the multiple alkyl groups present that provide increased stability for transition states resulting in highly efficient elimination processes.
C. Other Reactions:
Grignard Reactions: Tertiary halogenoalkanes are often utilized as starting materials in Grignard reactions due to their increased reactivity, which facilitates organomagnesium compounds formation.
Cross-Coupling Reactions: Halogenoalkanes can all be employed as cross-coupling partners to form new carbon-carbon bonds in cross-coupling reactions, leading to the production of complex organic molecules.
Understanding the roles played by primary, secondary, and tertiary halogenoalkanes in specific reactions allows chemists to select appropriate substrates and optimize reaction conditions efficiently in order to produce desired products efficiently.
Halogenoalkane reactivity depends upon their structural features such as steric hindrance or stability of intermediates formed during reactions.
Environmental impact and safety considerations
Halogenoalkanes, like any chemical compound, pose potential environmental and safety impacts that must be considered when handling, using and disposing of them responsibly in order to limit adverse impacts.
Here are some environmental and safety considerations associated with halogenoalkanes:
A. Environmental Impact:
Persistence: Chlorofluorocarbons (CFCs) and halons have been Identified as Contributing to ozone depletion by Depleting Earth’s protective ozone layer – Protecting Humanity against harmful UV Radiation.
Global Warming Potential: Hydrochlorofluorocarbons (HCFCs) and hydrofluorocarbons (HFCs), along with some halogenoalkanes such as hydrofluorocarbons (HFCs) are powerful greenhouse gases with high global warming potential, contributing significantly to climate change through their emissions, which cause global warming effects and contribute to climate change through climate change itself.
B. Toxicity and Health Hazards: These chemicals pose various hazards including potential toxicity issues that pose threats as well as potential toxicity issues when released from their sources of origins;
Human Health Effects: Halogenoalkanes may have serious negative health impacts that range from acute to chronic. Prolonged or excessive exposure could potentially have neurological, reproductive and developmental ramifications.
Environmental Toxicity: Halogenoalkanes that contain chlorine or bromine may be hazardous to aquatic life and other organisms in their immediate environments, with long-term impacts to ecosystems lingering as bioaccumulates through food chains.
C. Regulatory Measures and Alternatives:
Regulation: Due to their environmental and health impacts, many halogenoalkanes are subject to regulations and restrictions. International agreements such as the Montreal Protocol have been put in place in order to phase out production and usage of these ozone-depleting substances.
Substitution: At times, efforts are undertaken to find safer alternatives to halogenoalkanes with reduced environmental impacts and better safety profiles; one such example would be using hydrofluoroalkanes (HFAs) instead of CFCs/HCFCs in certain applications.
D. Safe Handling and Disposal:
Storage and Handling: Halogenoalkanes should be stored and handled according to safety guidelines and regulations, using proper ventilation, personal protective equipment (PPE), and containment measures to limit exposure and avoid accidents.
Waste Management: Halogenoalkanes should be disposed of following prescribed protocols. Recycling or appropriate disposal methods should be utilized to reduce environmental contamination.
Researchers, industries and users of halogenoalkanes must be cognizant of their potential environmental and safety impacts when working with these compounds. Responsible usage, adhering to regulations, and considering safer alternatives are all vital for minimizing environmental footprint and safeguarding human health and ecosystem wellbeing.
Potential future developments in the field
Halogenoalkane research and technology advances are constantly shifting the field forward, leading to further advances in organic synthesis, environmental considerations, and emerging technologies. Here are some possible developments within this space:
Green and Sustainable Synthesis: There has been a recent surge in interest for developing more eco-friendly synthetic methods for halogenoalkanes. This includes exploring alternative reaction conditions, catalytic systems, and renewable starting materials in order to lower their environmental footprint during production processes.
Replace Ozone-Depleting and High-GWP Compounds: Our efforts will continue to focus on replacing CFCs and halons that deplete ozone with safer alternatives, and research on low global warming potential alternatives that contribute to climate change mitigation.
Innovation of Efficient and Selective Reactions: Research will continue to seek improvements in the efficiency and selectivity of reactions involving halogenoalkanes, including developing novel catalysts, reaction conditions, or methodologies in order to increase yield and specificity of desired products.
Cross-Coupling Reactions: Cross-coupling reactions involving halogenoalkanes will see further advances, with researchers exploring new coupling partners, reaction conditions and catalysts that increase carbon-carbon bond formation for complex organic molecules. This will expand both their scope and efficiency for creating carbon bonds allowing more effective carbon bond synthesis processes to take place more easily.
Functionalization of Halogenoalkanes: Research will focus on creating new methods for selective functionalization of halogenoalkanes, introducing various functional groups into alkyl chains to produce tailored compounds with specific properties and applications.
Halogenoalkanes will play an essential role in medicinal chemistry for the synthesis of pharmaceuticals. Further effort will be undertaken to optimize their properties as potential drug candidates by optimizing reactivity, toxicity and pharmacokinetic properties.
Integration with Emerging Technologies: Halogenoalkane research will converge with emerging technologies like flow chemistry, artificial intelligence-driven synthesis and continuous manufacturing, creating opportunities for improved efficiency, reaction control and scaling during halogenoalkane synthesis.
Safety and Environmental Considerations: Ongoing research will investigate the environmental fate and impacts of halogenoalkanes as well as devising safer handling, storage, and disposal methods. This may involve exploring alternative solvents or cleaner technologies as well as improved waste management strategies.
As this field advances, interdisciplinary collaborations and technological innovations will lead to more sustainable, efficient, and safer processes involving halogenoalkanes that expand their applications across industries while meeting environmental concerns.
Conclusion
Understanding the differences among primary, secondary, and tertiary halogenoalkanes is fundamental in organic chemistry. Each compound demonstrates distinct properties that impact various reactions – with primary halogenoalkanes showing high nucleophilic substitution reactions while secondary and tertiary ones possessing variable degrees of reactivity due to steric hindrance or stability of carbocation intermediates.