Introduction of Finkelstein and Swarts Reaction
The Finkelstein reaction and Swarts reaction are two influential transformations in organic chemistry that involve substituting halogen atoms for carbon in organic molecules. Both processes play a vital role in synthesizing organic molecules, offering various advantages and challenges along the way.
The Finkelstein reaction, named in honor of Hans Finkelstein, involves exchanging chlorine or bromine for iodine in an organic molecule via nucleophilic substitution and is frequently conducted using polar solvents with silver salts for extra efficiency and versatility. The reaction has become one of the premier means of producing organic iodides due to its versatility and reactivity benefits.
Frederik Swarts first discovered the Swarts reaction, which involves replacing one or more chlorine or bromine atoms with fluorine atoms via radical-based reactions that employ strong Lewis acids to generate radicals which then react with fluorine gas to form fluorine compounds. This radical-based reaction serves as an important tool in fluorine chemistry by providing access to organic molecules with fluorine-containing elements.
Organic chemists need to understand the differences between these reactions in order to select the most appropriate methodology for any given synthesis goal. While the Finkelstein reaction is preferred for iodination processes and Swarts for fluorination reactions, each has its own set of advantages, limitations, and preferred reaction conditions. By doing so, we can gain a thorough knowledge of these essential halogen substitution reactions as well as their unique roles in organic synthesis.
Definition of Finkelstein Reaction
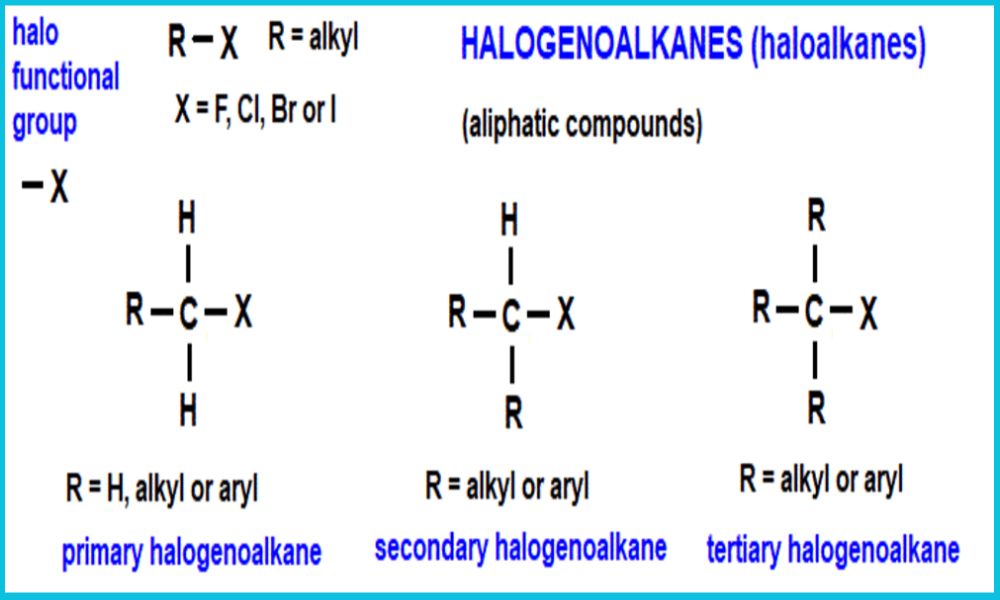
The Finkelstein reaction is a chemical process in which an alkyl or aryl halide compound is converted to another halide compound by swapping out one halogen atom (typically chlorine or bromine) with another (usually iodine). It was first described by Russian chemist Hans Finkelstein and first described publicly during the early 20th century.
The Finkelstein reaction occurs via a nucleophilic substitution mechanism where an iodide ion (in this instance) replaces any chloride or bromide groups attached to carbon atoms with its own iodide ion counterpart, typically taking place in polar and aprotic solvents such as acetone or dimethyl sulfoxide (DMSO), with assistance provided by adding silver salts such as AgNO3 that help remove less reactive silver halides precipitates (AgNO3).
The Finkelstein reaction has wide application in organic synthesis, especially when selective halogen exchange is desired. Iodine often provides more reactive and versatile substituents compared to other halogens; as a result, this method has been employed to prepare various organic iodides. Furthermore, its use has been employed in producing pharmaceuticals, natural products, and functional organic compounds.
Definition of Swarts Reaction
The Swarts reaction is a chemical process in which one or more halogen atoms in an organic compound are exchanged for another type of halogen atom; typically chlorine or bromine are exchanged for fluorine; it was named after Dutch chemist Frederik Swarts who discovered it in early 20th century.
The Swarts reaction works via a radical mechanism. A strong Lewis acid such as antimony pentachloride or pentafluoride converts halogen atoms to radicals which then react with fluorine gas (F2) molecules to form the desired product: organic molecules with fluorine atoms.
Due to the highly reactive and volatile properties of fluorine gas, the Swarts reaction must take place under highly rigorous conditions such as high temperatures and anhydrous environments. Nonpolar solvents like carbon tetrachloride (CCl4) or chloroform (CHCl3) are often employed during this process.
The Swarts reaction is an important tool in fluorine chemistry and has numerous applications in synthesizing fluorinated organic compounds for use in pharmaceuticals, agrochemicals, and materials science applications.
Fluorinated organic compounds often possess unique properties which make them beneficial in fields like medicinal chemistry and materials science; however, due to fluorine’s reactivity and potential side reactions the Swarts reaction requires careful control of reaction conditions and reagent selection for optimal results.
Comparison Table of Finkelstein and Swarts Reaction
Sure! Here’s a comparison table highlighting the key differences between the Finkelstein reaction and the Swarts reaction:
| Aspect | Finkelstein Reaction | Swarts Reaction |
|---|---|---|
| Type of Reaction | Nucleophilic Substitution | Radical Substitution |
| Halogen Substitution | Typically chlorine or bromine to iodine | Typically chlorine or bromine to fluorine |
| Mechanism | Nucleophilic substitution | Radical substitution |
| Key Reagents | Organic halide, iodide source, silver salt | Organic halide, fluorine gas, Lewis acid |
| Reaction Conditions | Polar, aprotic solvent | Nonpolar solvent, high temperature, anhydrous conditions |
| Stereochemistry | Retention of configuration | Retention of configuration |
| Selectivity | Good selectivity for iodination | Selective for fluorination |
| Applications | Preparation of organic iodides | Introduction of fluorine atoms into organic compounds |
| Limitations | Limited to chlorine and bromine | Harsh reaction conditions, potential for side reactions |
| Advantages | Versatile and reactive iodine species | Valuable for the introduction of fluorine atoms |
| Comparative Preference | Preferred for iodination processes | Preferred for fluorination processes |
Please note that this table provides a general overview of the key differences between the two reactions. There may be specific variations and considerations depending on the reaction conditions, reagents, and substrates used.
Importance and applications of these reactions in organic chemistry
Both the Finkelstein reaction and Swarts reaction play key roles in organic chemistry and play an instrumental role in synthesizing organic compounds. Here are their significance and key applications:
Importance of Finkelstein Reaction:
Iodination: The Finkelstein reaction is widely utilized for the selective replacement of chlorine or bromine atoms with iodine atoms in organic molecules, creating valuable intermediates used in pharmaceutical, agrochemical, and materials synthesis processes.
This process results in organic iodides which serve as valuable intermediates in such areas as pharmaceutical synthesis, agrochemical production, as well as material creation processes.
Reactivity and Versatility: Iodine-substituted compounds often exhibit enhanced reactivity and diverse chemical behavior compared to their halogenated counterparts, making the Finkelstein reaction an efficient method for adding iodine, thus giving access to unique reactivity patterns as well as functional group transformations.
Applications of Finkelstein Reaction:
Pharmaceutical Synthesis: The Finkelstein reaction is widely utilized for pharmaceutical synthesis. Iodine-substituted molecules may exhibit altered pharmacokinetic properties, improved binding affinity, or enhanced stability making this reaction invaluable in medicinal chemistry.
Materials Science: Organic iodides can be used in the synthesis of liquid crystals, dyes, and polymers using the Finkelstein reaction. Iodine adds physical or chemical properties that improve materials such as liquid crystals. Swarts Reaction is equally essential.
Fluorination: The Swarts reaction is an essential process for adding fluorine atoms into organic molecules. Fluorine replacement often leads to significant modifications of molecular properties, including lipophilicity increases, metabolic stability increases, and biological activity enhancement. Being able to selectively and efficiently fluorinate organic molecules thus becomes of immense value in pharmaceutical and agrochemical industries.
Fluorine Chemistry: Fluorine is widely known for its unique potency in organic reactions. Through the Swarts reaction, fluorinated compounds are synthesized which have applications across a range of fields including materials science, the petrochemical industry, and medicinal chemistry.
Applications of Swarts Reaction:
Medicinal Chemistry: The Swarts reaction allows the incorporation of fluorine atoms into drug molecules, yielding compounds with enhanced pharmacological properties. Fluorinated drugs often exhibit improved bioavailability, metabolic stability, and receptor affinity; making the Swarts reaction essential in creating fluorinated pharmaceuticals.
Materials and Specialty Chemicals: Fluorinated organic compounds possess many outstanding characteristics, including thermal stability, chemical inertness, and unique solubilization properties. The Swarts reaction provides an efficient means of synthesizing fluorinated intermediates used as building blocks in specialty chemicals, polymers, and advanced materials production.
Finkelstein and Swarts’s reactions play key roles in organic chemistry by providing efficient methods for halogen substitution. Their applications span pharmaceuticals, agrochemicals, materials, and specialty chemicals where adding iodine or fluorine atoms impart desirable properties to target molecules.
Understanding and making use of these reactions expands one’s synthetic toolbox and enables novel compounds with tailored properties for specific uses to be created.
Substitution pattern and stereochemistry of the products
Substitution Pattern and Stereochemistry of Finkelstein Reaction Products:
Finkelstein reaction products vary based on reaction conditions and starting organic halide structure; typically however, this reaction results in the substitution of one chlorine or bromine atom with one iodine atom.
Finkelstein reactions usually result in a simple one-for-one substitution pattern. For example, starting organic halides such as primary alkyl chlorides or bromides react with an iodide source to form their respective primary alkyl iodides; similarly, secondary alkyl halides will generate secondary alkyl iodides as their end products.
Finkelstein reactions generally exhibit retention of configuration in terms of stereochemistry, so if an organic halide that starts as optically active is converted to optically inactive products via the S N 2 mechanism and nucleophilic substitution with inversion of configuration occurs; however, other factors, such as steric hindrance or nearby functional groups may alter its outcome and alter the final stereochemistry.
Substitution Pattern and Stereochemistry in Swarts Reaction:
In the Swarts reaction, its substitution pattern and stereochemistry of products are affected by radical-based mechanisms and the incorporation of fluorine atoms.
Concerning its substitution pattern, Swarts reactions generally involve replacing chlorine or bromine atoms with fluorine atoms resulting in organic chlorides or bromides being transformed into fluorides and vice versa.
Swarts reaction typically leads to retention of configuration, as it utilizes radical reactions for its catalysis. While stereochemistry of starting organic halides may often remain intact in their product form, radical reactions may sometimes result in racemization or loss of stereochemical integrity depending on whether there are neighboring chiral centers present or the radical intermediate undergoes rearrangement during their course of reactions.
Noteworthy is the fact that both Finkelstein and Swarts reactions can be affected by numerous factors that can alter their stereochemistry, such as starting halide structure, reaction conditions, and any neighboring functional groups or steric effects. Therefore it is crucial to take this into account when predicting or analyzing stereochemical profiles for these reactions.
Limitations and challenges associated with the Finkelstein reaction
Finkelstein reaction is an invaluable method for organic synthesis involving halogen exchange; however, it also presents several limitations and challenges which must be considered carefully before proceeding with its use.
These restrictions and obstacles include:
Finkelstein Reaction for Chlorine and Bromine Only: The Finkelstein reaction is designed for use only when replacing chlorine and bromine atoms with iodine; it may not be applicable when exchanging other halogens such as fluorine or replacing fluorine atoms with other halogens such as chlorine and bromine atoms.
Reactivity and Selectivity: The Finkelstein reaction can vary in its reactivity and selectivity depending on which halogens are exchanged and their position in an organic halide structure, such as highly hindered substrates with multiple halogen atoms or highly hindered organic halides containing multiple halogen atoms exhibiting lessened selectivity or reactivity.
Side Reactions: In some instances, Finkelstein reactions can result in side reactions such as elimination or rearrangement processes that reduce yield or create undesirable byproducts. These side reactions could decrease product yield or lead to unexpected byproduct formation.
Utilization of Silver Salts: Finkelstein reactions require the addition of silver salts such as silver nitrate to accelerate the removal of less reactive halide ions as silver halide precipitates, but their addition adds both cost and complexity to the setup of reactions.
Solubility and Reaction Conditions: Choosing a solvent and reaction conditions that allow for maximum solubility for reactants and products as well as controlling reaction rate and selectivity is of key importance in producing desired products that may otherwise require special handling or different conditions for production.
For instance, certain substrates or desired products may have limited solubility in certain solvents or need special handling or alternative conditions in order to achieve maximum solubility for production.
Moisture Sensitivity: Moisture can disrupt the Finkelstein reaction and hinder its progress, hydrolyzing organic halides or interfering with its reactions. Therefore, to maintain anhydrous conditions during setup and handling of reagents.
Limitations in Stereochemistry: While Finkelstein reactions typically proceed without loss of configuration, certain factors such as nearby functional groups or steric hindrance could have an impactful effect on their stereochemistry.
Note that many of these limitations and challenges can be minimized through careful optimization of reaction conditions, substrate selection and selection of suitable reagents. Even with its inherent limitations, the Finkelstein reaction remains an invaluable method for halogen exchange, particularly for synthesizing organic iodides.
Limitations and challenges associated with the Swarts reaction
The Swarts reaction, while valuable for adding fluorine atoms into organic compounds, does have some limitations and challenges that must be considered before using this technique. These constraints include:
Harsh Reaction Conditions: To achieve optimal results with the Swarts reaction, harsh conditions including high temperatures and anhydrous environments must be present to facilitate the generation and reactivity of halogen radicals. Unfortunately, such harsh reactions can restrict its application to specific substrates or even lead to unintended side reactions.
Selectivity Issues: The Swarts reaction can sometimes experience selectivity problems due to generated halogen radicals reacting with other functional groups within a molecule and producing undesirable side products. Achieving fluorination efficiently may prove challenging when multiple reactive sites exist within one complex molecule.
Safety Issues: Fluorine gas poses significant safety concerns due to its high reactivity and toxicity, necessitating careful handling and storage in order to ensure it does not pose health risks, necessitating additional measures and precautions to be taken against exposure.
Potential Side Reactions: Due to its radical-based nature, Swarts reaction may produce side reactions such as homolytic aromatic substitution or undesired radical reactions that result in complex reaction mixtures and reduce yield and selectivity for fluorinated products. These side reactions could further diminish yield and selectivity by creating complex reactions with complex reaction mixtures resulting from side reactions that arise as side reactions from radical-based processes.
Limitations on Substrate Compatibility: Not all organic halides are suitable substrates for the Swarts reaction, as certain functional groups or structural features in starting halides may hinder radical generation or subsequent fluorination processes, leading to reduced reactivity or yields.
Synthetic Challenges: Fluorine can significantly change the properties and reactivity of organic molecules, necessitating additional synthetic steps in order to safely handle fluorinated products as well as address potential reactivity or stability concerns associated with fluorine-containing compounds.
Stereochemistry Considerations: The Swarts reaction operating through radical intermediates may present difficulties when it comes to maintaining stereochemical integrity. Radical reactions may lead to racemization or loss of stereochemical information when dealing with neighboring chiral centers or rearrangement-prone structures nearby.
Though limited and complex, the Swarts reaction remains an effective method for adding fluorine atoms into organic compounds. By optimizing reaction conditions and substrate selection as well as taking appropriate safety measures, many of its inherent limitations can be managed and effective fluorination can occur efficiently and selectively.
Variations in substitution patterns and stereochemistry of the products
Variations in Substitution Patterns in Finkelstein Reaction:
Although Finkelstein reactions generally involve replacing chlorine or bromine atoms with an iodine atom, their substitution patterns can differ depending on the structure of starting organic halides. Some examples include:
Simple Substitution: The Finkelstein reaction usually leads to a simple one-for-one substitution where chlorine or bromine atoms are exchanged for an iodine atom, producing one product with an unambiguous substitution pattern.
Multiple Substitutions: When starting with an organic halide with multiple halogen atoms, Finkelstein reactions can result in multiple substitutions; multiple iodine atoms will replace their original counterparts and produce compounds with multiple iodine substituents.
Sequential Substitutions: When performing the Finkelstein reaction on a compound containing multiple halogen atoms, sequential substitution can occur instead of simultaneous one – instead one halogen is first replaced by iodine before proceeding with further substitution steps in subsequent steps.
Variations in Stereochemistry during Finkelstein Reaction:
In Finkelstein reactions, starting organic halides’ stereochemistry may influence the final product; however, its stereochemical outcome is typically determined by the nucleophilic substitution mechanism S N 2 in play during this reaction. Examples of variations include:
Retention of Configuration: The Finkelstein reaction tends to proceed without loss of configuration, meaning if an organic halide starting material is optically active and chiral, its resultant product usually retains that same stereochemistry.
Inversion of Configuration: Although retention of configuration is typically the goal, Finkelstein reactions may lead to inversion in certain cases, particularly if there are neighboring functional groups or steric hindrances that impede reaction pathways.
Racemization: Under certain conditions or substrates that favor racemization, Finkelstein reactions can result in the breakdown of stereochemical integrity, producing racemic mixtures as an outcome.
When studying or predicting the stereochemical outcome of a Finkelstein reaction, its products’ stereochemistry depends on many variables such as starting halide structure, reaction conditions, and neighboring functional groups or steric effects. Therefore it is crucial to take these aspects into account and conduct appropriate stereochemical analysis when studying or predicting its outcome.
Specific applications where one reaction is preferred over the other
Although both the Finkelstein reaction and Swarts reaction offer distinct advantages and applications, depending on your desired transformation and starting materials one may be preferred over the other. Here are a few examples:
Finkelstein Reaction Is Best Used:
Iodination: For organic molecules requiring an introduction of an iodine atom, the Finkelstein reaction provides outstanding selectivity for iodination processes that allow for the creation of organic iodides.
Preparation of Iodinated Pharmaceuticals: The Finkelstein reaction is commonly employed in the production of iodinated pharmaceutical compounds, where its properties include altered pharmacokinetic properties, improved binding affinity, or enhanced stability – making the Finkelstein reaction an indispensable asset in medicinal chemistry.
Introduction of Iodine for Materials Synthesis: The Finkelstein reaction finds applications in materials science where the introduction of iodine is desired. Organic iodides may serve as intermediates during liquid crystal, dye, and polymer synthesis processes and impart particular physical or chemical properties into their final products.
Preferred Use of Swarts Reaction:
Fluorination: When it comes to fluorinating organic compounds, the Swarts reaction is the way of choice for adding fluorine atoms. The Swarts reaction allows for the selective substitution of chlorine or bromine for fluorine in order to obtain fluorinated organic compounds.
Development of Fluorinated Pharmaceuticals: The Swarts reaction has proven its worth in developing fluorinated pharmaceuticals. Fluorine substitution can significantly increase the bioavailability, metabolic stability, and receptor affinity of drug molecules; making the Swarts reaction an integral component of medicinal chemistry.
Specialty Chemicals and Materials: The Swarts reaction has become an indispensable tool in the production of specialty fluorine-containing organic compounds, providing unique properties such as thermal stability, chemical inertness, and specific solubilization behavior. Fluorinated organic compounds find widespread application across materials science, petrochemistry, and polymer applications.
When selecting between Finkelstein and Swarts reactions for specific applications, it is essential to take several factors into account, including material reactivity, desired properties of final products, reaction conditions compatibility, and desired transformations involving different halogen substitutions.
Furthermore, both reactions can sometimes be employed together in sequence to meet desired transformation goals.
Future prospects and potential advancements in these reactions
Future possibilities and advancements of Finkelstein reaction and Swarts reaction can be seen across many areas of organic synthesis and methodology development, including:
Development of Catalysts: Catalyst development represents one of the greatest advancements for both Finkelstein and Swarts reactions, offering efficient and selective catalysts that improve reaction rates, selectivity, milder conditions, and expand the substrate scope.
Future Advancements may include expanding the substrate scope for both reactions, to permit substitution of various organic halides. By developing methodologies enabling the exchange or specific substitution of other halogens (for instance fluorine-to-chlorine or chlorine-to-bromine), more versatile synthetic opportunities may become possible.
Green and Sustainable Approaches: As environmental considerations play a greater role in organic synthesis, future advances may include creating more eco-friendly versions of Finkelstein and Swarts reactions – such as alternative solvents or reduced or eliminated toxic or costly reagent requirements; waste generation could also be minimized through sustainable approaches.
Application in Flow Chemistry: Applying continuous flow systems to Finkelstein and Swarts reactions may offer several advantages, including enhanced reaction control, greater safety features, and scalability. Future improvements may focus on optimizing these reactions for flow conditions to ensure efficient production of iodinated and fluorinated compounds.
Mechanistic Understanding: Advancements in computational chemistry and experimental techniques will contribute to an increased understanding of the Finkelstein and Swarts reactions’ mechanisms, reaction pathways, selectivity factors, and reactivity factors influencing these reactions, thus helping create more efficient and reliable conditions for their reactions.
Future Innovations may include the integration of Finkelstein and Swarts reactions with other halogen exchange methodologies to form integrated strategies that allow for the selective exchange of different halogen atoms within one molecule – thus providing access to tailored compounds with tailored properties.
These advances could significantly enhance both Finkelstein and Swarts reactions, expanding their synthetic utility, improving reaction efficiency, and creating novel halogenated compounds with applications in pharmaceuticals, materials science, and other areas of research.
Conclusion
Finkelstein and Swarts reactions are two crucial tools in organic chemistry for halogen substitution. The Finkelstein reaction can be used for replacing chlorine or bromine with iodine for selective, versatile organic iodide production; on the other hand, Swarts allows fluorine atoms to enter by replacing chlorine or bromine atoms with fluorine.
Both reactions possess distinct advantages, such as selectively adding certain halogens into organic compounds for synthesizing compounds with specific properties. They also present challenges, including side reactions and specific conditions required for reaction; as well as safety considerations associated with specific reagents.